 Specific gravity can be measured in any unit.ĭoes temperature affect the density of air and water? What is the unit of measurement for specific gravity? The unit of measurement for density is kg/m³. What is the unit of measurement for density? 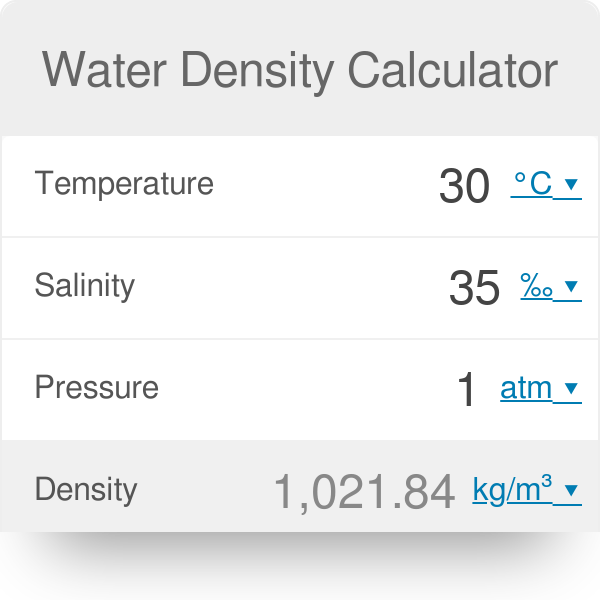
Specific gravity is used to compare the densities of different substances. 
Specific gravity is the ratio of a substance’s density to that of a reference substance, usually water.Īir has a specific gravity of around 0.0012. Water is denser than air because its molecules are closer together and have a higher mass per unit volume. The density of water at standard conditions is around 1000 kg/m³. 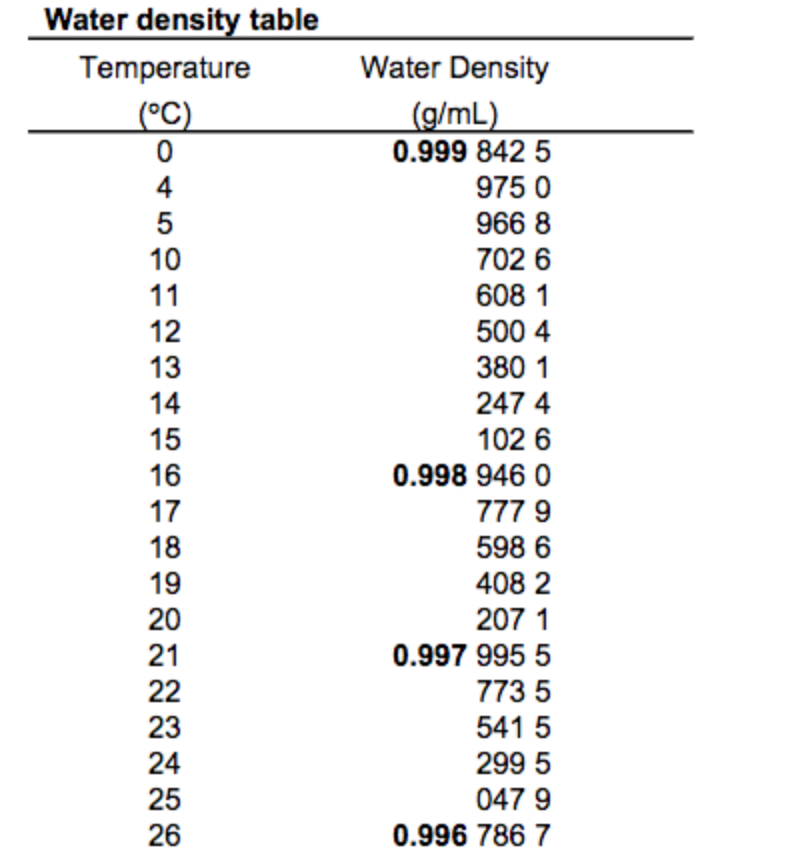
The density of air at standard conditions is around 1.225 kg/m³. Water is approximately 800 times denser than air. What is the difference between their densities? This is why humid air feels lighter than dry air, even though the total mass of the air is the same. As the humidity levels increase, the density of air decreases, since water vapor has a lower density than dry air. Moreover, the density of air is also affected by the presence of water vapor. However, as the temperature increases, the density of air decreases, and as the pressure increases, the density of air also increases. However, the density of air varies with temperature, pressure, and humidity levels.įor example, at a temperature of 20☌ and atmospheric pressure, the density of dry air is approximately 1.2041 kg/m3. Air is primarily composed of nitrogen (78%), oxygen (21%), and small amounts of other gases, such as carbon dioxide, argon, and neon.Īt standard temperature and pressure (STP), which is defined as 0☌ (273.15 K) and 101.325 kPa, the density of air is approximately 1.225 kg/m3. The density of air is defined as the mass per unit volume, usually measured in kilograms per cubic meter (kg/m3) or grams per cubic centimeter (g/cm3). This difference in density is why water has much more resistance to movement than air and why it can support heavier objects. This means that water is about 800 times denser than air. At standard temperature and pressure (STP), air has a density of about 1.225 kg/m³, while water has a density of 1000 kg/m³. Density of Air vs WaterĪir has a much lower density than water. In this article, we will explore the density of air and water and compare their properties, including their effects on the environment and the impact on human health. In particular, the density of air and water has important implications in a wide range of applications, from meteorology to marine engineering.Īir is a gaseous substance, while water is a liquid, and the difference in their density is substantial. It plays a crucial role in many areas of science and engineering, including fluid mechanics, thermodynamics, and materials science. = 1000 g/L (exactly) = 1000 kg/m 3 (exactly) ≈ 62.4280 lb/cu ft (approximately) ≈ 133.5265 oz/US gal (approximately)ġ lb/cu ft ≈ 0.01601846 g/cm 3 (approximately)ġ oz/US gal ≈ 0.Density is a fundamental physical property of matter, defined as the amount of mass per unit of volume. 
The density of water is about 1 g/cm 3, since the gram was originally defined as the mass of one cubic centimetre of water at its maximum density at 4 ☌ (39 ☏). It is equivalent to the units gram per millilitre (g/mL) and kilogram per litre (kg/L). It is defined by dividing the CGS unit of mass, the gram, by the CGS unit of volume, the cubic centimetre. The gram per cubic centimetre is a unit of density in the CGS system, and is commonly used in chemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
